Describe What Surface Tension Is Using Kinetic-molecular Theory
The macroscopic phenomena of pressure can be explained in terms of the kinetic molecular theory of gases. Use the editor to format your answer Paint Question 23 Calculate the molality of the following solution.

Kinetic Molecular Theory And Liquids And Solids Ppt Video Online Download
Surface tension allows needles and paper clips to float in water if placed carefully on the surface.

. KPI 11 Describe using particulate diagrams the different states of matter solid liquid gas and plasma 12 describe the kinetic molecular theory of particles 13 List the five assumptions of the kinetic molecular theory of particles 14 use the kinetic molecular theory to explain the physical properties of gases with respect to expansion fluidity low density compressibility and. Select all that apply. According to the basic assumption of kinetic molecular theory gas particles.
Surface tension resists penetration of objects into a liquid. Have a significant volume with respect to the volume of the container they occupy c. What is surface tension.
The kinetic molecular theory of matter states that. This in turn determines whether the substance exists in the solid liquid or gaseous state. It also explains why drop of water are spherical in shaped 22.
Lose kinetic energy when colliding 4. 4What does the negative slope of waters solidliquid equilibrium line indicate. The same process is responsible for the concave.
The kinetic-molecular theory explains the physical properties of solids liquids and gases in terms of the energy of particles and the. Define an ideal gas. Describe the Gas state according to the kinetic-molecular theory.
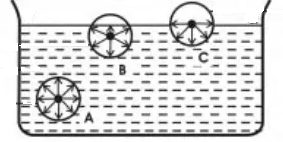
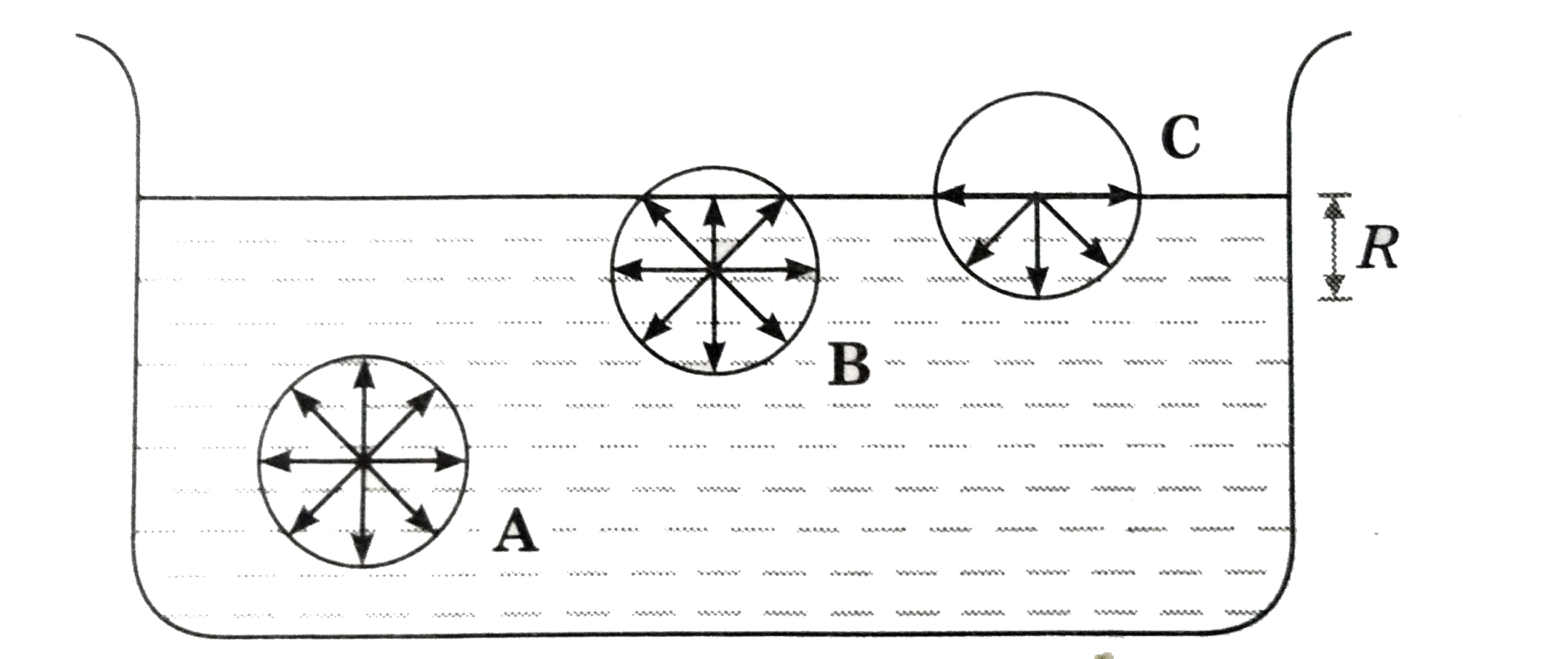
How do molecular forces affect surface tension. ADescribe all the phase changes a sample of solid water would undergo when heated to its critical temperature at a pressure of 100 atm. These intermolecular forces tend to pull the molecules into the liquid and cause the surface to tighten like an elastic film or skin.
The Kinetic Molecular Theory KMT is a model based on a series of postulates that explain the behavior of matter. Using gas kinetic molecular theory show that under typical atmospheric conditions of pressure and temperature corresponding to an altitude of 5 km see Appendix V collisional deactivation of a CO 2 molecule will be much faster than reemission of the absorbed radiation. Why is carbon dioxides positive.
Gas particles are in constant line motion so they move until they make contact with a wall of a container. Check all that apply The particles of a liquid have enough kinetic energy to move randomly past each other allowing the liquid to flow. Take the collision diameter to be 0456 nm and the radiative lifetime of the 15-μm band of CO 2 to be 074 s.
Describe kinetic molecular theory. Pressure and KMT. Surface tension is the energy required to increase the surface area of a liquid by a unit amount and varies greatly from liquid to liquid based on the nature of the intermolecular forces eg water with hydrogen bonds has a surface tension of 729 x 10-2 Jm 2 at 20C while mercury with metallic bonds has as surface tension that is 15.
4- Liquids exhibit surface tension a force that tends to pull adjacent parts of a liquids surface together thereby decreasing the surface area to its smallest possible size. All particles have energy but the energy varies depending on the temperature the sample of matter is in. Up to 24 cash back Properties of Liquids and the Kinetic-Molecular Theory continued Surface Tension continued Capillary action is the attraction of the surface of a liquid to the surface of a solid.
Values of the surface tension and surface excess internal energy of a number of simple liquids are obtained by numerical integration of the Kirkwood-Buff-Fowler equations. Which of the following options correctly describe the phases of matter in terms of kinetic molecular theory. All calculations are based on the Lennard-Jones potential with parameters which are known to reproduce the thermodynamic properties of the bulk liquids.
Identify each statement as True or False. Describe the motion of gas particles in an ideal gas. Use the kinetic theory of matter to describe the properties of matter.
Describe Kinetic Molecular Theory. Select all that apply. Attractive forces of the molecules create surface tension.
Using the kinetic molecular theory explain why a gas can be easily compressed while a liquid and a solid cannot. Move rapidly in a constant random motion d. This attraction tends to pull the liquid molecules upward along the surface and against the pull of gravity.
37g of glucose C6H1205 in 64g of water Use the editor to format your answer. Are far apart b. Matter is made up of particles that are constantly moving.
Explore KMT including its postulates and learn about the properties of solids. Concept Notes 101 Ch 101 Reading Assessment. Surface tension a force that tends to pull adjacent parts of a liquids surface together thereby decreasing surface area to the smallest possible size capillary action.
Assume the case in which a gas molecule represented by a sphere is in a box length L Figure 1Through using the assumptions laid out above and considering the sphere is only moving in the x-direction we can examine the. Explain the difference between an ideal gas and a real gas and under what conditions they exist. Use the editor to format your answer 1 Point Question 3 Describe the 4 standards that gases need to have in order to fit the kinetic molecular theory of gases.
The force that pulls the outer layer of a liquids surface together reducing its surface area to the smallest possible size Describe the liquid state according to the kinetic-molecular theory. 5- Liquids have capillary action the attraction of the surface of a liquid to the surface of a solid.

Chapter 12 Chemistry Vocabulary Flashcards Quizlet

Ch 10 Kinetic Molecular Theory Notes

The Kinetic Molecular Theory Of Gases Ppt Download

The Kinetic Molecular Theory Of Gases Ppt Download

Pdf Molecular Theory Of Surface Tension

The Kinetic Molecular Theory Of Gases Ppt Download

Explain Surface Tension On The Basis Of Molecular Theory Brainly In

The Kinetic Molecular Theory Of Liquids Solids Ppt Video Online Download

Kinetic Molecular Theory Of Liquids State Of Matter Liquid Chemistry Digital Kemistry Youtube

Solved 1 State The Five Assumptions Of Kinetic Molecular Theory 2 Write The Formula For Ke Of Moving Object And What Does The Kinetic Energy And Temperature Of A Specific Gas Depend On

Kinetic Molecular Theory Intermolecular Forces

Chemistry Kinetic Molecular Theory States Of Matter Chapter Ppt Download
Surface Tension The Concept Its Characteristics And Factors Affecting It

The Kinetic Molecular Theory Of Liquids Solids
Explain The Phenomenon Of Surface Tension On The Basis Molecular Theory

Kinetic Molecular Theory Of Matter Chemistry Flashcards Quizlet

The Kinetic Molecular Theory Of Liquids Solids


Comments
Post a Comment